31 Tellurium is a Group 6A element in Period 5 What would you expect for the valence-shell configuration of tellurium A 5s25p4B 5s25p6C 6s26p4D 6s26p632 Thallium has the ground-state configuration Xe4
- B. 5s 2 5p 6
- B. It is a d-transition element.
- C. group 2A, period 4
- C. sp3
- A. paramagnetic
- A. P-O
- D. HBr
- D. PbCl2
- B. 55.67
- B. diamagnetic
II. Short answers and calculations (4 × 5 points)
a. Br-Br b. H-S-H c. F-N=F d. Se-Se
- H+(aq) + Br-(aq) + NH3(aq) → NH4+(aq) + Br-(aq)
- 2H+(aq) + 2Cl-(aq) + Ba2+(aq) + 2OH-(aq) → 2H2O(l) + Ba2+(aq) + 2Cl-(aq)
- Pb2+(aq) + 2Br-(aq) → PbBr2(s)
- MgCO3(s) + H+(aq) → Mg2+(aq) + H2O(l) + CO2(g)
-
Using the ideal gas law: PV = nRT n = (PV)/(RT) n = (786 mmHg)(0.25 L) / [(0.08206 L atm/mol K)(394 K)] n = 0.027 mol Molecular weight = (2.56 g) / (0.027 mol) = 94.81 g/mol
-
ΔH = [2(ΔHf CO2) + 2(ΔHf H2O)] - [ΔHf C2H4 + 3(ΔHf O2)] + [2(ΔHf CO2) + 3(ΔHf H2O)] - [ΔHf C2H6 + 7/2(ΔHf O2)] ΔH = [2(-393.5) + 2(-285.8)] - [-1411] + [2(-393.5) + 3(-285.8)] - [-1560 + 7/2(-286)] ΔH = -136.2 kJ
-
To determine the limiting reactant, we need to calculate the number of moles of NaClO that could be obtained from each reactant: From NaOH: 2 mol NaClO/mol NaOH x 1.44 mol NaOH = 2.88 mol NaClO From Cl2: 1 mol NaClO/mol Cl2 x 1.47 mol Cl2 = 1.47 mol NaClO Therefore, Cl2 is the limiting reactant. The number of moles of NaClO that could be obtained is 1.47 mol
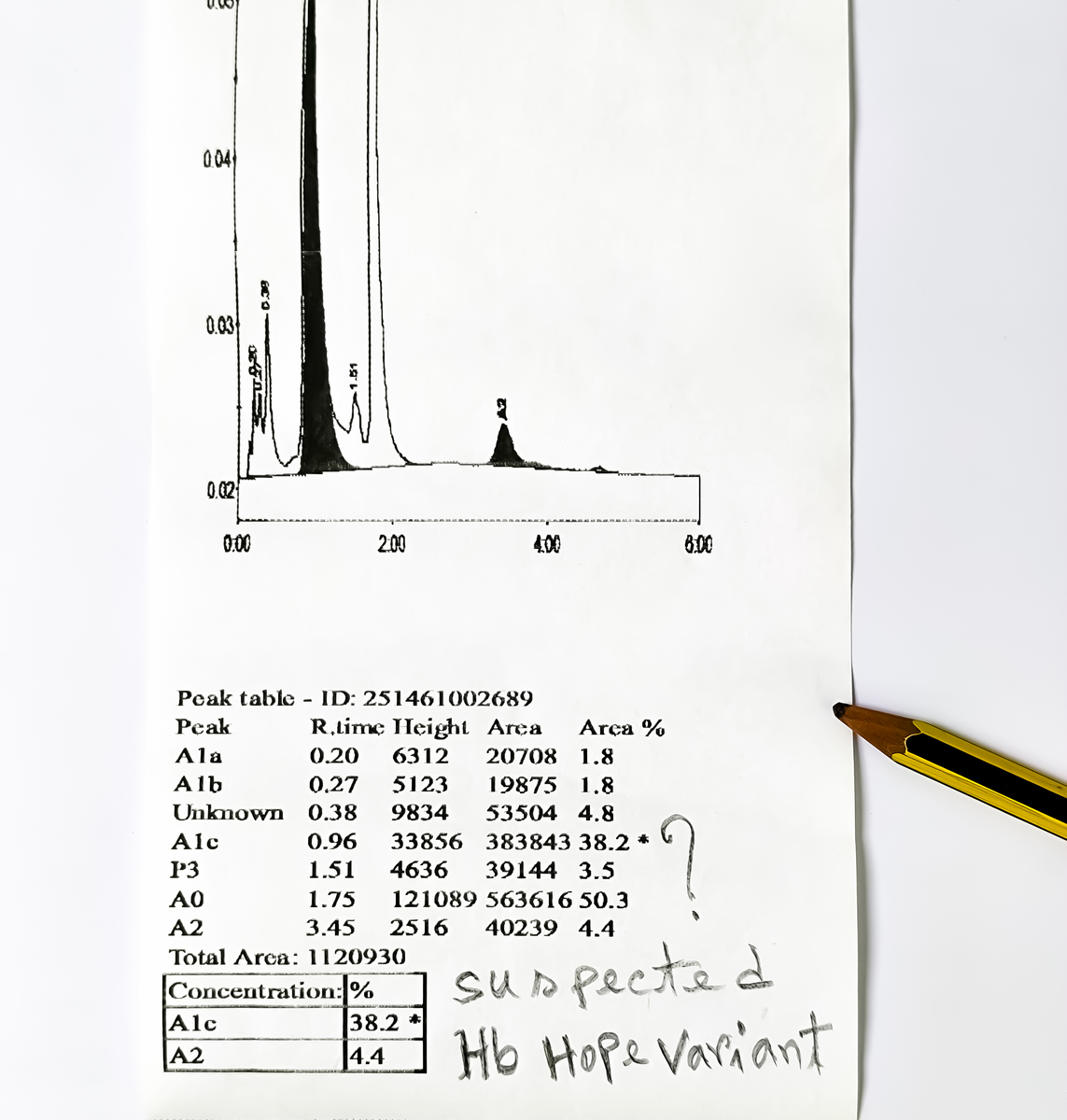
原文地址: https://www.cveoy.top/t/topic/gH63 著作权归作者所有。请勿转载和采集!