please revise The main problem is that the clinical trial’s result is not very good We checked the clinical data that was submitted to FDA The non-superity hypothesis is not established The non-inferi
The main issue at hand is that the clinical trial's results are not satisfactory. After reviewing the clinical data submitted to the FDA, we found that the non-superiority hypothesis was not established, with the non-inferior value set at 15% and the point value at 18%. This indicates a high likelihood of trial failure, which is likely why the FDA has not yet approved it. Additionally, the NMPA's review process is even stricter than that of the FDA, making it difficult to obtain approval. While there are exceptions, such as the Impella left cardiac assist system, which showed effectiveness in a specific group during subgroup analysis and after extending the trial period to 90 days, these cases are rare. Therefore, we suggest that the data be re-analyzed to determine any potential improvements that could be made to increase the likelihood of approval
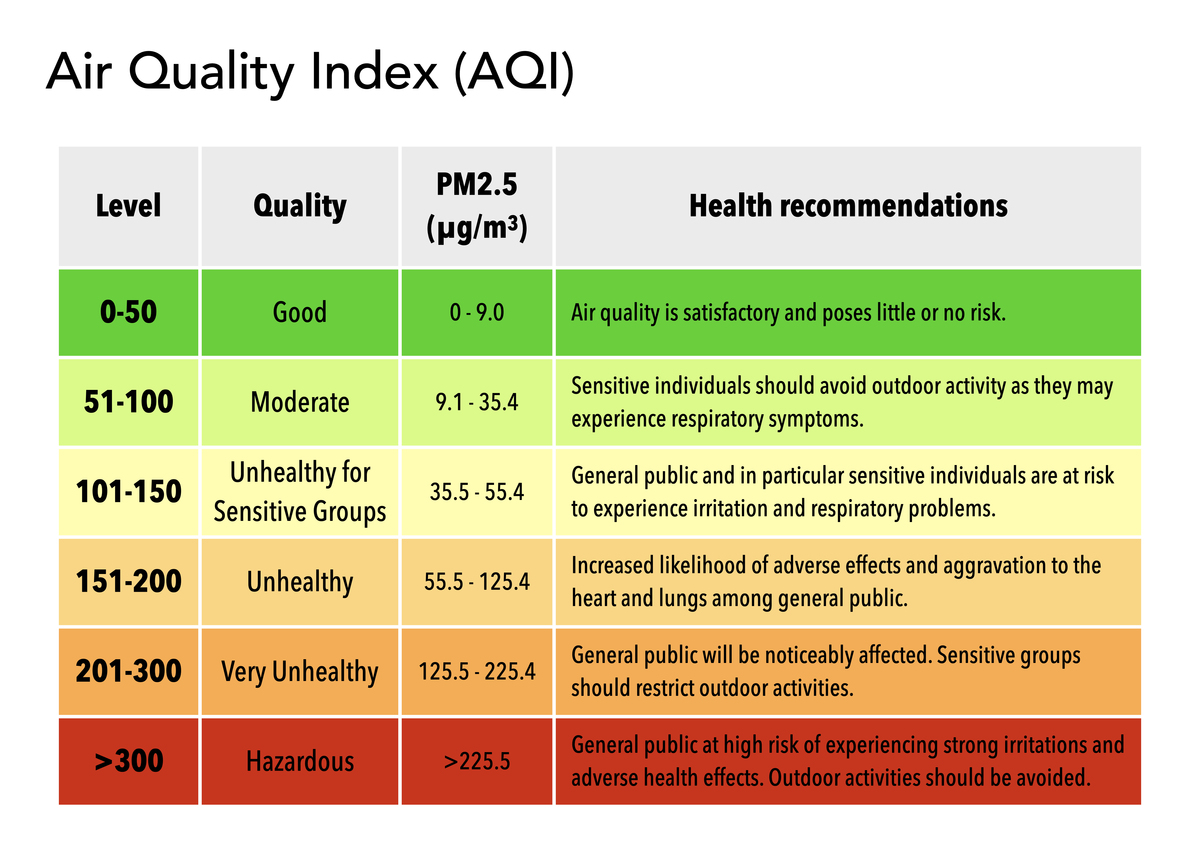
原文地址: http://www.cveoy.top/t/topic/hunt 著作权归作者所有。请勿转载和采集!